At LABS, we are dedicated to advancing medical AI by integrating machine learning with medical imaging analysis, multi-organ and multi-omics computational biology, and population genetics. While the era of AI-driven precision medicine holds immense promise, our research specifically addresses the critical hurdles of reproducibility, disease heterogeneity, and multi-scale modeling to ensure these innovations successfully transition into clinical practice.
For LABS' long-term mission, please refer to Dr. Wen's recent Nature Biomedical Engineering invited Comment: Towards a multi-organ, multi-omics medical digital twin
Traditional AI/ML approaches often model organs in isolation, but biological aging is a systemic process. By integrating multi-organ imaging with multi-omics data, spanning genomics, proteomics, and metabolomics, we can capture the complex "cross-talk" between biological systems.
This holistic approach allows us to move beyond chronological age to identify organ-specific biological clocks and disease subtypes . By mapping the genetic architecture of these systems, we can pinpoint how dysfunction in one organ (such as the heart or metabolic system) accelerates decline in others (like the brain). Ultimately, this multi-scale modeling is essential for precision medicine, enabling us to predict individual disease trajectories and identify therapeutic targets long before clinical symptoms emerge.
Our work in this area has been featured in leading journals including Nature, Nature Medicine, Nature Aging, Nature Biomedical Engineering, and Nature Mental Health.
Neurodegenerative and psychiatric disorders are profoundly complex, often defying simple clinical categorization. Our research leverages AI-derived endophenotypes to bridge the gap between gross neuroimaging observations and underlying biological etiology. By treating these high-dimensional imaging traits as quantitative markers, we apply population genetics and computational biology to uncover the specific genomic loci that govern brain structure and function.
This integration allows us to deconstruct disease heterogeneity, identifying the precise molecular pathways that drive individual pathological trajectories. By linking imaging "signatures" directly to genetic risk, we transform descriptive neuroimaging into a tool for mechanistic discovery, moving us closer to biologically-informed diagnostics and targeted interventions.
Our work in brain imaging genetics and AI endophenotypes are featured in PNAS, JAMA Psychiatry, and Nature Biomedical Engineering.
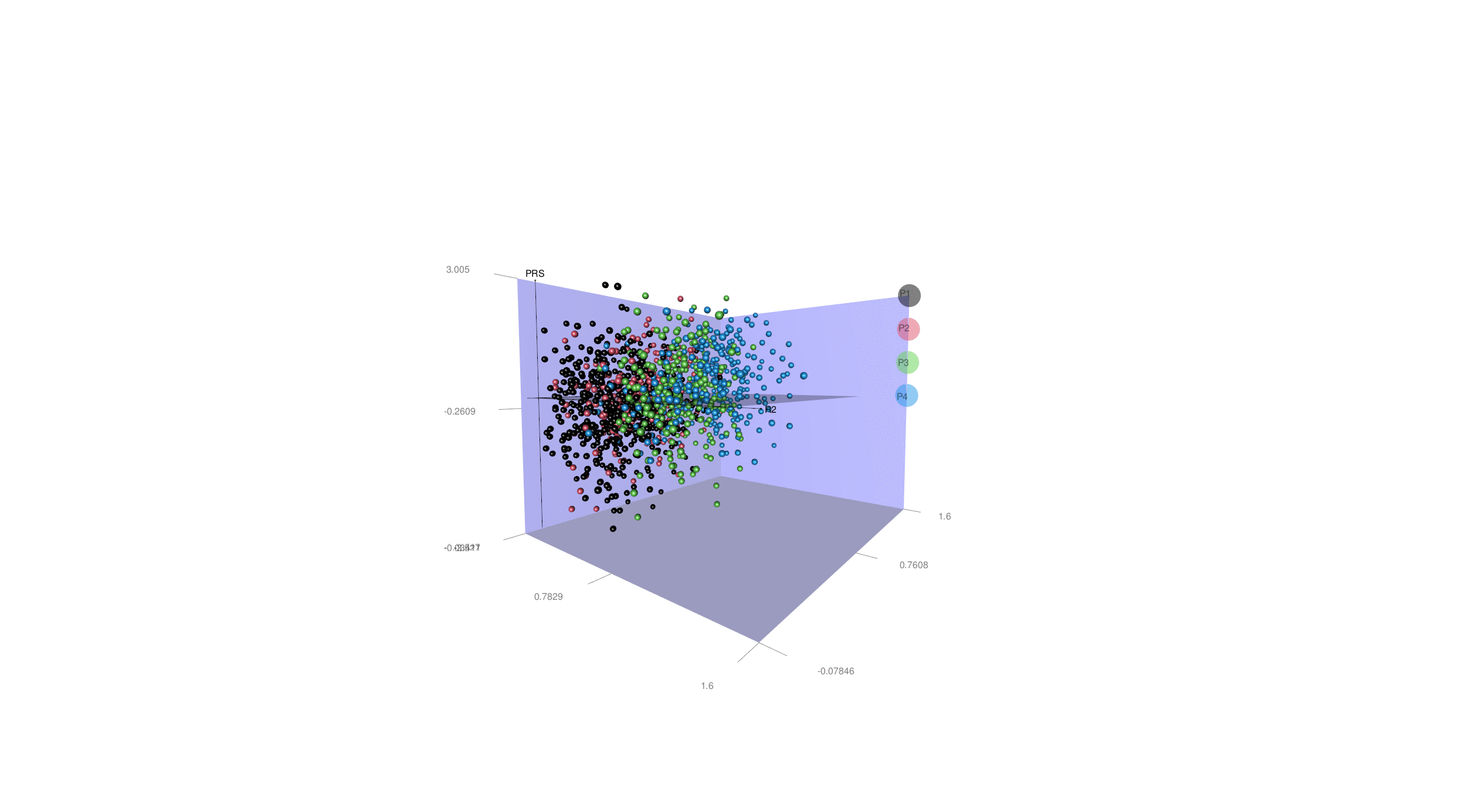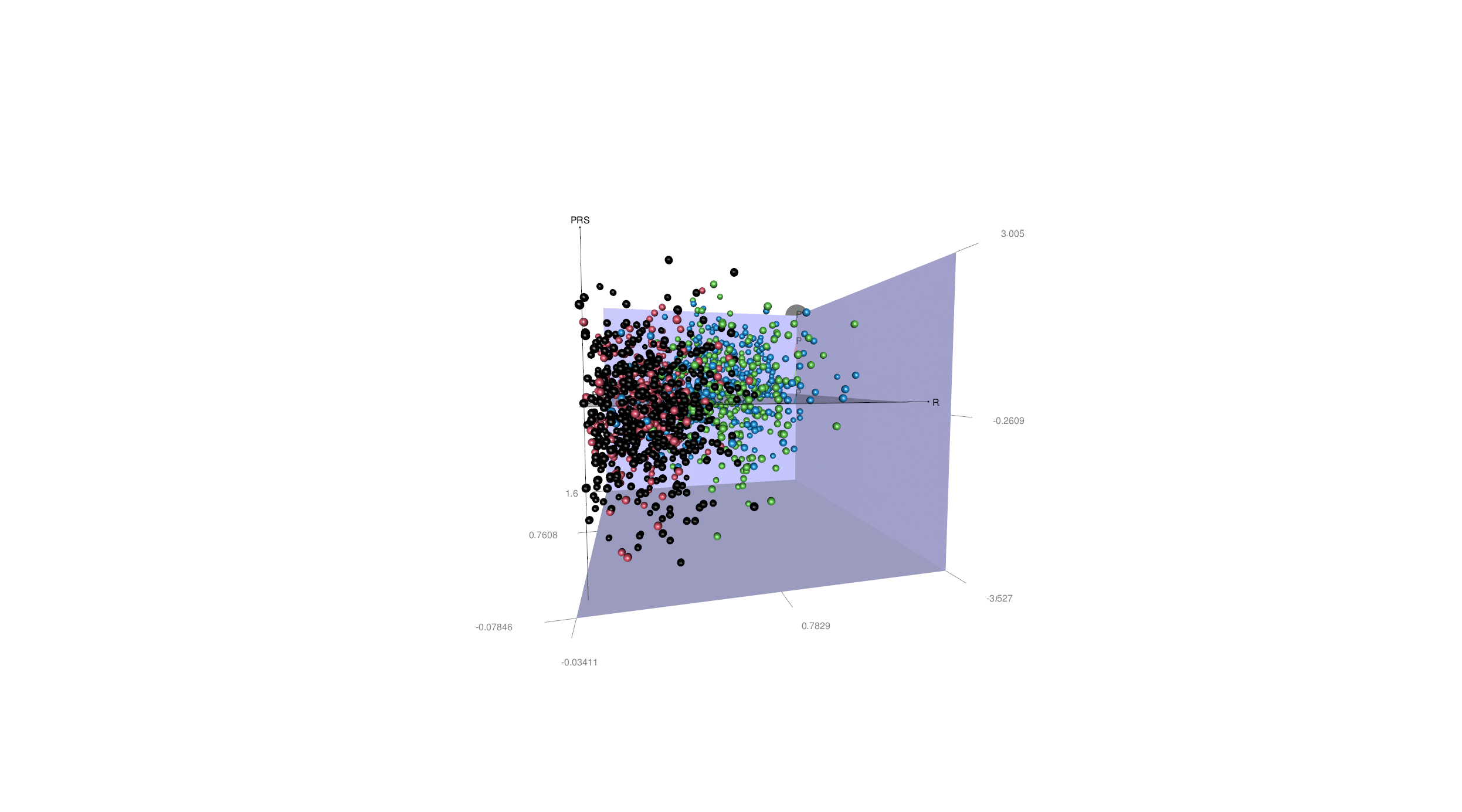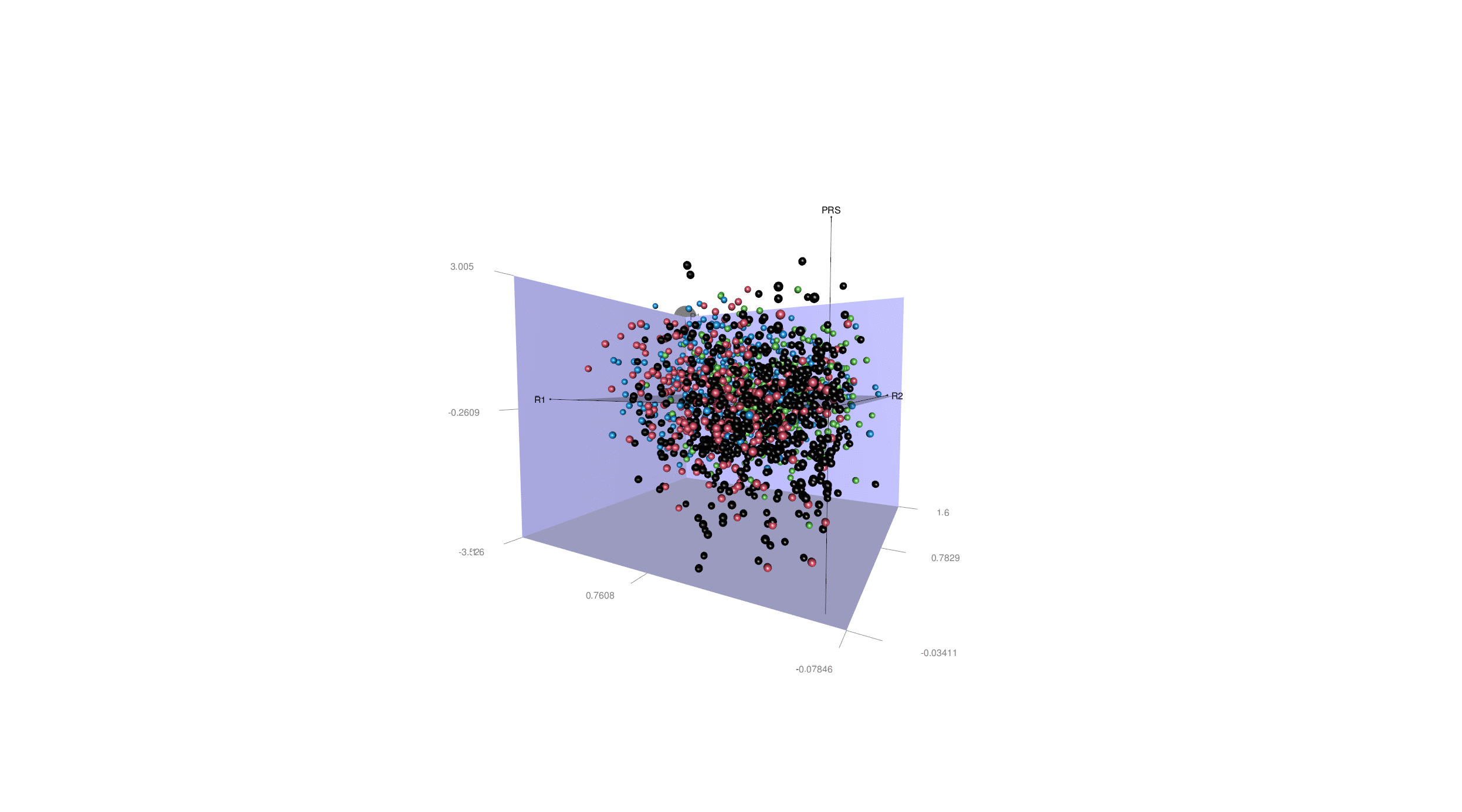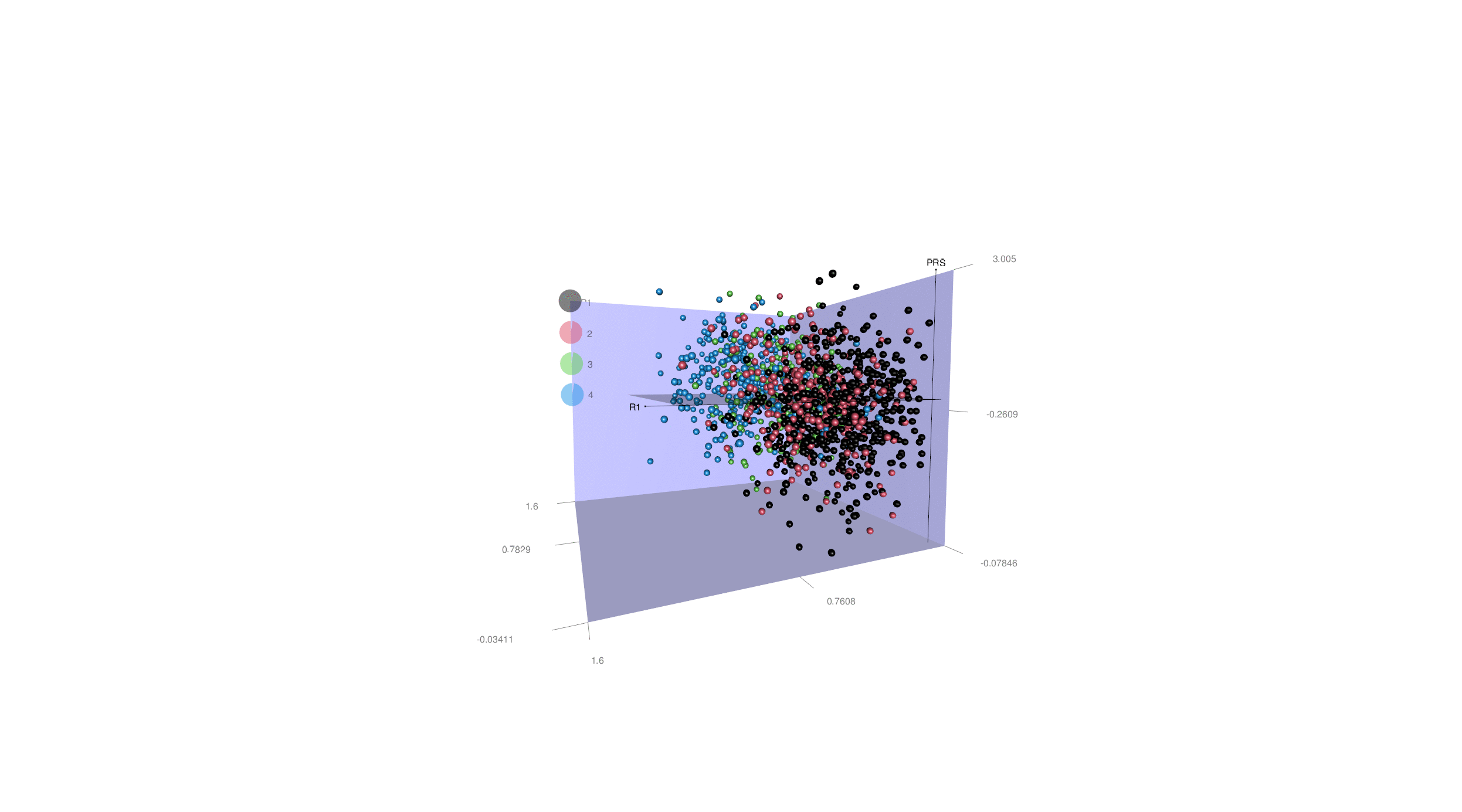
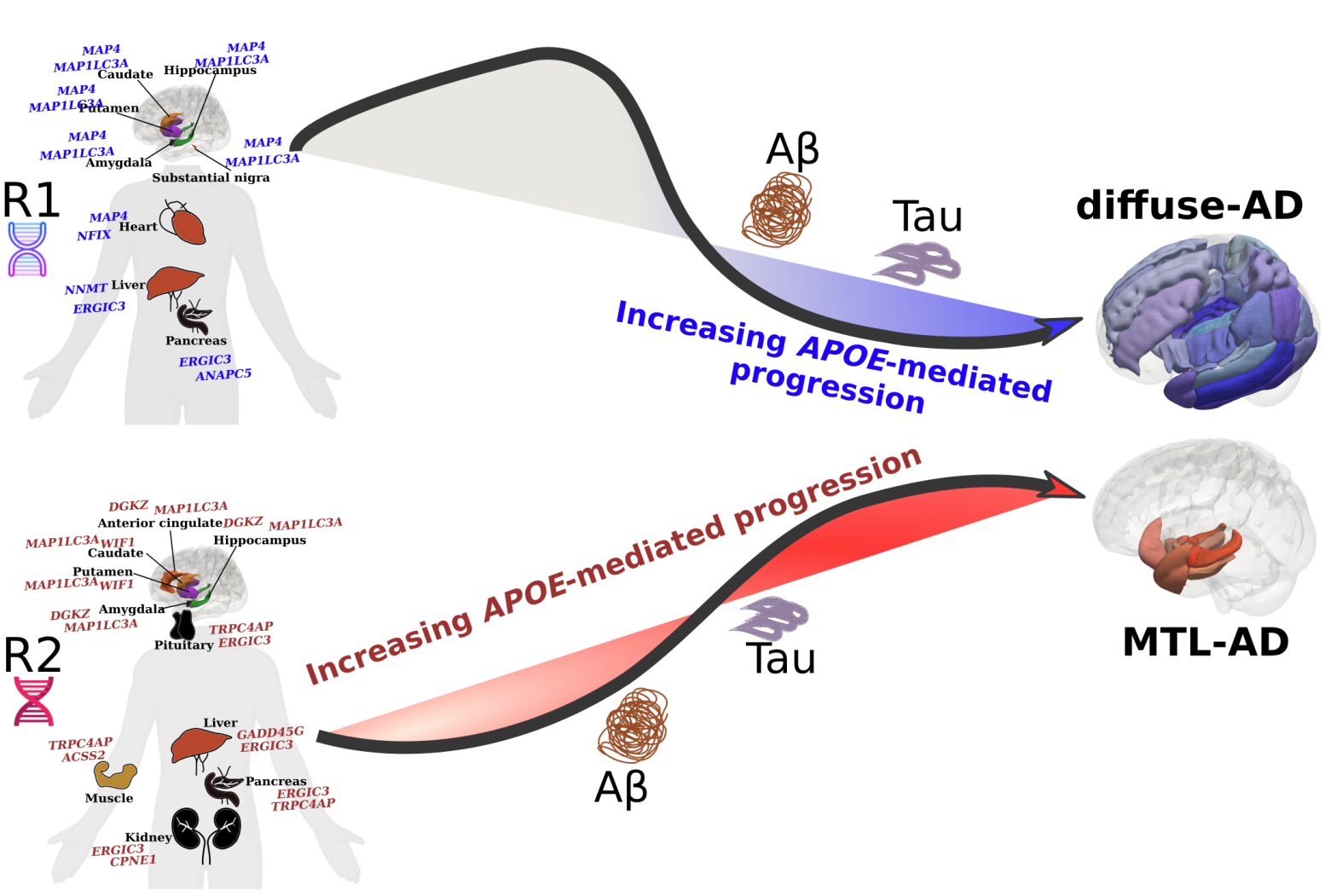
A major barrier to precision medicine is the vast phenotypic and genetic heterogeneity within traditional diagnostic categories. Patients sharing the same clinical diagnosis, such as Alzheimer’s disease (AD), schizophrenia, or autism spectrum disorder (ASD), often exhibit vastly different neuroanatomical and clinical trajectories.
Our lab develops and applies clustering and deep learning frameworks to dissect this complexity. By mapping individual patients into high-dimensional representations, we identify distinct disease subtypes/dimensions and individualized susceptibility profiles. This data-driven stratification — as illustrated in our work on AD dimensions (R1/R2) — allows us to move beyond "one-size-fits-all" models toward personalized prognostic and therapeutic strategies.
Our research on disease subtyping and heterogeneity has been published in Nature Communications, Medical Image Analysis, JAMA Psychiatry, and top-tier AI conferences including ICLR and MICCAI.

The clinical translation of AI for Alzheimer’s disease (AD) is often hindered by a lack of objective benchmarking and systemic issues like data leakage. Our research group pioneered open-source frameworks that provide the scientific community with standardized pipelines to evaluate machine learning (ML) and deep learning (DL) models across diverse neuroimaging modalities, including T1w MRI, diffusion MRI, and PET.
By critically assessing the performance of various architectures — from linear SVMs to complex CNNs — we have established rigorous benchmarks that prioritize model transparency and reproducibility. Our publicly available software and findings serve as a cornerstone for the field, ensuring that future AI innovations are built on a foundation of scientific integrity and are truly "clinical-ready."
Our highly cited methodological contributions and software frameworks are featured in Medical Image Analysis, NeuroImage, Bioinformatics, and Neuroinformatics.